-
ABOUT US
-
R&D
-
AGORA
-
PR
-
RECRUITMENT
-
CONTACT
AGORA
2024-2027
3H Bio timeline for Business and Anti-Obesity Medicine Development
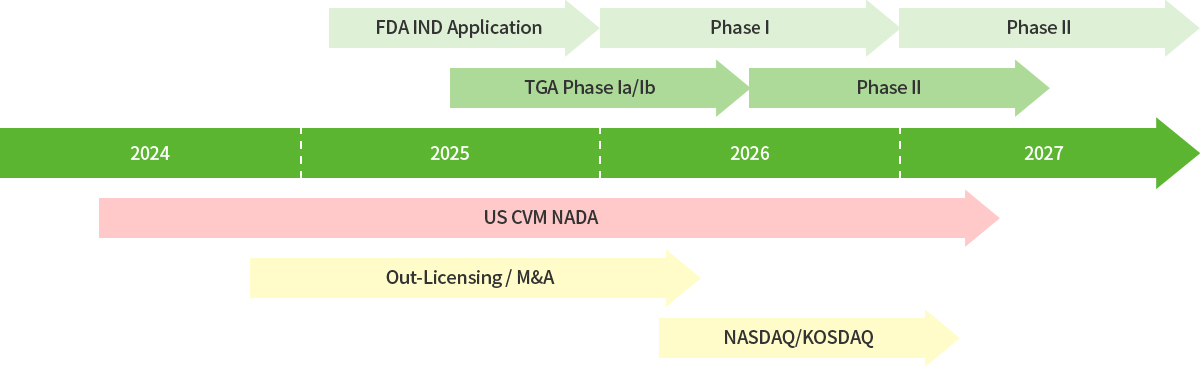
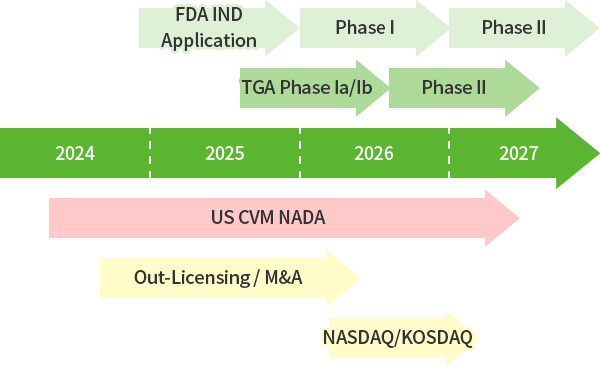
-2025. Oct: US FDA IND Application
-2026. Apr: Report of Clinical Phase 1a Study in Australia
-2026. Sep: Report of Clinical Phase 1b Study in Australia
Clinical 3HOTP - FDA IND (CBER) development process
| Task | 2025 | 2026 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ~June | July | Aug | Sep | Oct | Nov | Dec | Jan | Feb | Mar | Apr | |||
| FDA | Engineering Batch |
|
|
|
|||||||||
| CTM Manufacture |
|
|
|
||||||||||
| Gap analysis for IND |
|
|
|
|
|||||||||
| Clinical protocol |
|
|
|
|
|
||||||||
| Pre-IND Activities |
|
|
|
||||||||||
| IND preparation & submission (Module) | M 1 |
|
|
||||||||||
| M 2 |
|
|
|
|
|||||||||
| M 3 |
|
|
|
||||||||||
| M 4&5 |
|
||||||||||||
| IND Submission | |||||||||||||
| Australia | IRB |
|
|
||||||||||
| Phase 1a |
|
|
|
|
|||||||||
| Phase 1b |
|
|
|
|
|||||||||





